|
This is due to the difference in electronegativity between carbon and oxygen atoms. It has carbon atom oxygen atom having 1 lone pairs of electrons on each atom.Ģ: What is the hybridisation of the carbon atom in CO?Ī: The carbon atom in CO undergoes Sp hybridisation to form 2 Sp hybrid orbitals.Ī: The bond angle in CO is 180 degree due to the linear arrangement of the 2 atoms in the molecule.Ī: Yes, CO has a dipole moment of 0.122 Debye. This makes Carbon monoxide molecule linear in shape.Ī: The Lewis structure of CO consists of a triple bond between the carbon atom and the oxygen atom. The bond angle in carbon monoxide (CO) is 180 degrees. This permits CO to dissolve in polar solvents making an important industrial gas used in a variety of processes. Dipole moment points from the carbon atom towards the oxygen atom. And the separation of charges creates a dipole moment in the molecule. This creates a partial negative charge on the O atom and a partial positive charge on the C atom. The shared pair of electron in the triple bond are more strongly attracted towards the oxygen atom. This means that oxygen has a greater ability to attract electrons towards itself in the CO molecule. The electronegativity of oxygen is higher than that of carbon. = 6 – 2 – 1/2 *6 =+1 Formal charge calculation in CO Lewis structure moment in CO Structure :Ĭarbon monoxide (CO) has a net dipole moment due to the difference in electronegativity between carbon and oxygen atoms. 
Formal charge calculation in CO lewis structure : This hybridisation explains the high reactivity of CO and its ability to act as a strong ligand in coordination compounds. Triple bond between Carbon and oxygen is shorter and stronger than a double bond but weaker than a true triple bond. And the remaining two 2p orbitals of the carbon atom form 2 pi bonds with the O atom.ĬO is a linear molecule with a bond angle of 180 degrees. Sp hybridised orbitals are oriented linearly to form sigma bond with the oxygen atom. The carbon atom hybridises its 2s and one of the 2p orbitals to form 2 Sp hybridised orbitals. So, CO forms triple bond to complete its octet.Ĭarbon atom undergoes Sp hybridisation that involves the promotion of one of its 2s electrons to the 2p orbital. In CO molecule, the carbon atom has 4 valence electrons and the oxygen has 6 valence electrons. Visit the link to understand the concept of CO Lewis structure Hybridisation in CO Lewis structure :Ĭarbon atom in carbon monoxide molecule undergoes Sp hybridisation. This movement makes carbon positively charged and oxygen negatively charged in the third resonating structure of ( CO ) You can learn the resonating structures of CO from the diagram. Resonating structures in CO Lewis structure more pi bond moves from double bond towards more electronegative O atom in the second resonating structure. This movement makes the negatively charged carbon and positively charged oxygen neutral in the 2nd resonating structure. 
In the first structure, one pi bond from triple bond move towards more electronegative O atom. See the resonating structures of CO below in the diagram: CO Lewis structure Resonating Structures of CO Lewis structure 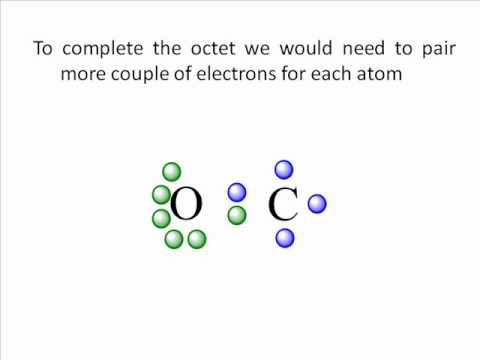
But rather they are delocalised throughout the molecule. But in resonating structures the electrons are not localised in specific bonds. Lewis structure represents the distribution of valence electrons in a molecule. CO Lewis structure indicates that the molecule is linear in shape having bond angle of 180 degrees. The triple bond in CO makes it a highly reactive molecule with some unique properties like its ability to bind to metal centres in coordination complexes. To complete the octets of both carbon and oxygen atoms they share a triple bond, consisting of one sigma bond and two pi bonds. CO Lewis structureĪs we know, carbon has four valence electrons, and oxygen has six valence electrons. Also, learn the bond angle, dipole moment according to CO Lewis structure molecular geometry and formal charges. We will also learn about the carbon monoxide formal charges and hybridisation. Here, we will discuss about CO Lewis structure molecular geometry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
